Get Healthy!
Staying informed is also a great way to stay healthy. Keep up-to-date with all the latest health news here.
07 Jul
Untreated High Cholesterol Causing Needless Heart Attacks and Stroke
In a new study, researchers estimate nearly 100,000 non-fatal heart attacks and up to 65,000 strokes could be avoided if people eligible for statins and other cholesterol-lowering drugs were taking them.
05 Jul
Teenage Drivers Spend About 21% of Each Road Trip on the Phone
Researchers surveyed more than 1,100 teenage drivers and found cell phones are a big distraction behind the wheel – even though many are aware of the danger to themselves and others.
02 Jul
Top 3 Medical Emergencies At School
A new study identifies three medical emergencies that account for the most EMS calls at schools. Researchers recommend improved training for these target areas.
U.S. Hospitals Face Shortages in Essential Staff Due to Foreign Residency Visa Delays
The Trump administration's recent travel and visa restrictions have left some hospitals in the United States without essential staff, according to a report from the Associated Press.
International doctors who were set to start their medical training this week are finding they are stumbling at the last hurdle -- a visa to enter the...
- Stephanie Brown HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
Six Key Traits That Make Someone 'Cool,' Study Finds
What does it take to be considered “cool”?
A new international study may have the answer — and it turns out cool people tend to act the same way no matter where they live.
Researchers from universities in Chile, the United States and beyond found that people seen as “cool” share six key personality...
- I. Edwards HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
Oregon Infants Sickened By Lead in Recalled Target Baby Food
MONDAY, July 7, 2025 — Two babies in Oregon got sick with lead poisoning after eating baby food from Target that was previously recalled, Oregon health officials said.
The food was part of the Good & Gather brand sold at Target stores and made by the company Fruselva. It was recalled back in March, but the Oregon Health Authority...
- I. Edwards HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
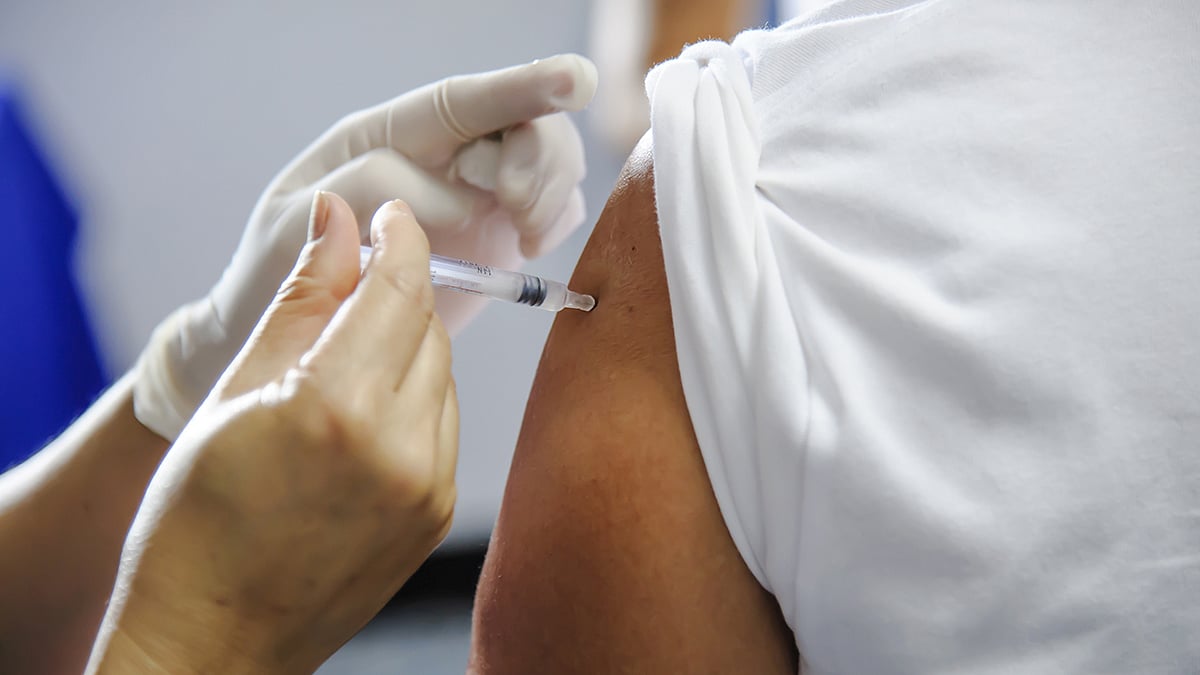
CDC Now Recommends RSV Vaccine for Some Adults 50+
Adults as young as 50 may now qualify for an RSV vaccine if they have certain health conditions, according to a quiet update from the U.S. Centers for Disease Control and Prevention (CDC).
This marks a change from the CDC’s previous recommendation, which only offered the shot to people 60 and older who were at high risk, The Asso...
- I. Edwards HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
Rheumatoid Arthritis Patients Benefit From Blood Flow-Restricted Strength Training
Folks who work out to ease their rheumatoid arthritis (RA) symptoms might find some relief from an unexpected source – a blood pressure cuff.
People who apply pneumatic cuffs to a limb while working out experience reduced pain and improved strength and physical performance, a small-scale study indicates.
This new form of exerci...
- Dennis Thompson HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
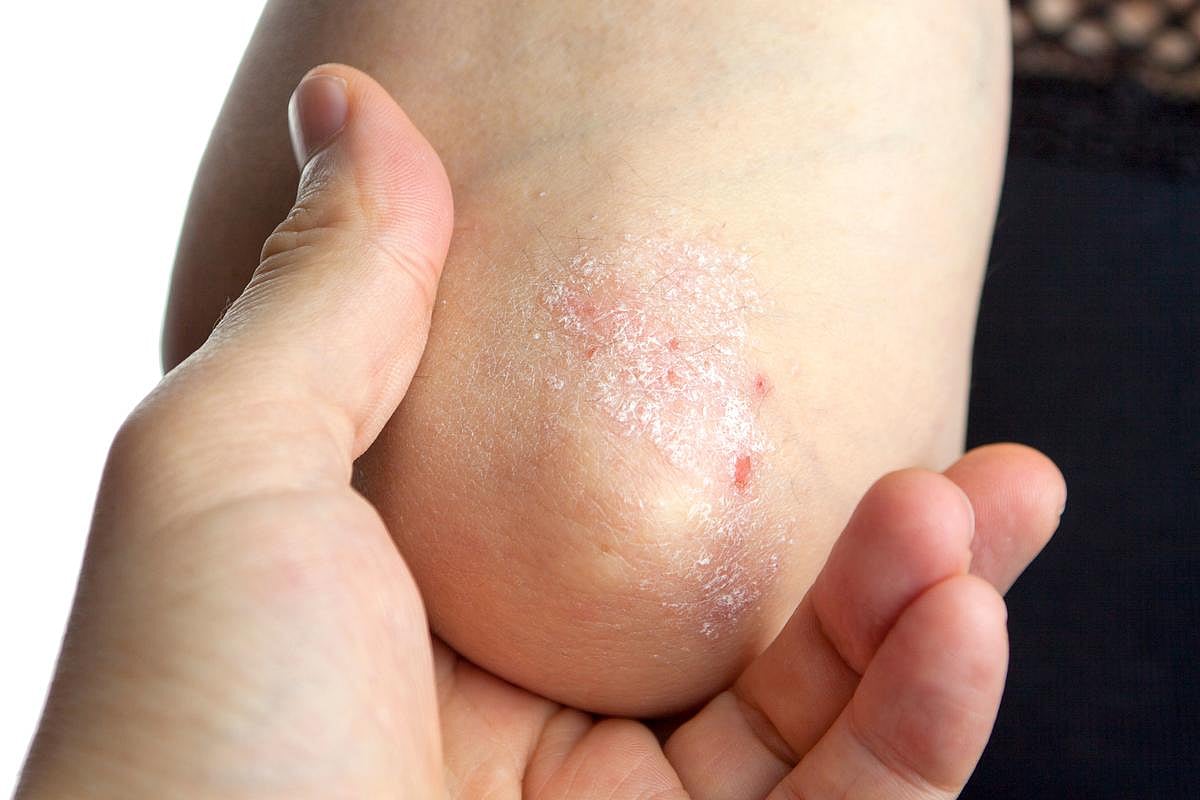
Psoriasis 101: Causes, Symptoms and Treatments, Explained
Psoriasis is a chronic autoimmune condition that mainly affects the skin. When you have psoriasis, your immune system sends signals to your skin cells to grow too fast.
Normally, skin cells take about a month to form and rise to the surface. In people with psoriasis, this process happens in just a few days. Because the skin can’t sh...
- Dr. Duaa AbdelHameid HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
More Than 14 Million Will Die Following U.S. Foreign Aid Cuts
More than 14 million children and adults will die from preventable causes during the next five years as a result of U.S. foreign aid cuts, a new study projects.
Programs supported by the U.S. Agency for International Development (USAID) prevented 91 million deaths between 2001 and 2021 in low- and middle-income countries, researchers repor...
- Dennis Thompson HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
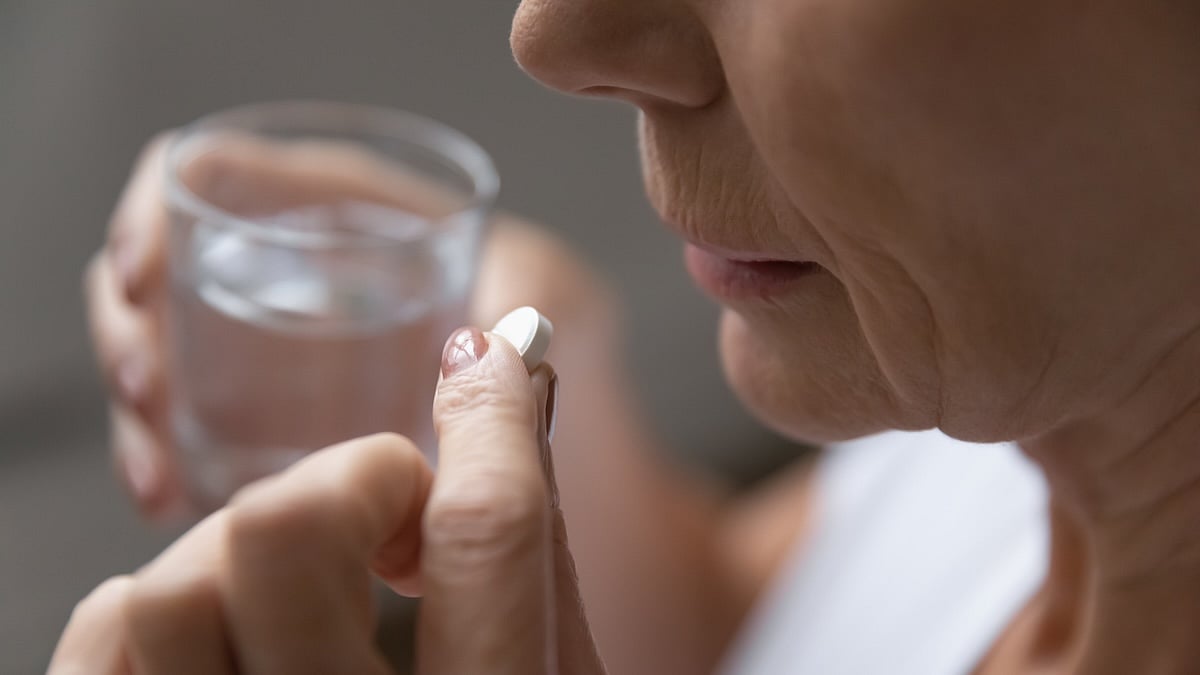
Many Older Adults Taking Daily Aspirin Despite Tighter Guidelines
About 1 in 6 older adults takes daily aspirin to prevent heart disease, even though stricter guidelines say it’s not for everyone, a new study says.
Worse, nearly a quarter of those folks started taking aspirin without a doctor’s recommendation, and about 1 in 10 haven’t discussed their use with any health care provider, ...
- Dennis Thompson HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
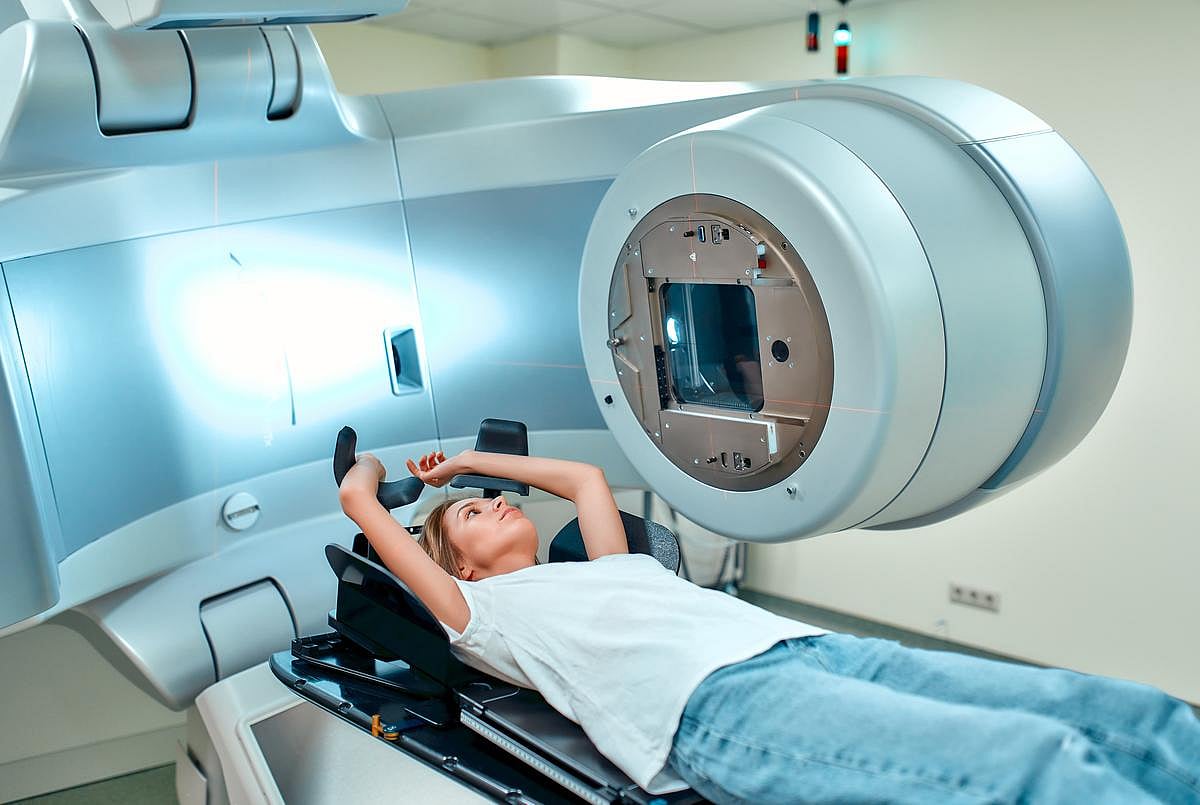
Is Radiation Therapy For Cancer a Double-Edged Sword?
Radiation therapy to treat cancer might be a double-edged sword that increases the risk posed by other malignant tumors, a new study argues.
While radiation is often used alone or in combination with other treatments to control cancer, high doses of radiation can promote the growth of metastatic tumors that weren’t directly treated w...
- Dennis Thompson HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
Type Of Hormone Therapy Can Alter Breast Cancer Risk, Study Says
Some women have expressed concerns about the risk of breast cancer associated with using hormone therapy to treat symptoms of menopause like hot flashes and night sweats, and now, new research suggests that one type of hormone therapy might increase your risk if you're younger than 55.
Women in this age group who were treated wit...
- Dennis Thompson HealthDay Reporter
- |
- July 7, 2025
- |
- Full Page
Screen Time May Slow Learning Skills, Study Finds
Screens are a part of daily life for many families — but too much screen time in the early years could slow a child’s language development, a new study suggests.
That’s because young children learn best when they can touch, explore and play with real objects, Sarah Kucker, a psychologist at Southern Methodist University i...
- I. Edwards HealthDay Reporter
- |
- July 6, 2025
- |
- Full Page
Is One Type of Water Healthier Than Another? Here’s What Experts Say
Alkaline. Electrolyte. Flavored. Walk down the beverage aisle and you’ll find all kinds of waters promising extra health perks. But are these fancy waters really better for you?
Not really, Tufts University experts say.
“There's no physiological basis that there's some metabolic benefit to these specialty waters over just...
- I. Edwards HealthDay Reporter
- |
- July 5, 2025
- |
- Full Page
Hearing Aids Are a Boon To Social Life, Study Finds
Some folks won’t use hearing aids because they’re worried the devices will make them look old or get in the way of their social life.
Nothing could be farther from the truth, a new evidence review says.
Hearing aids dramatically improve a person’s social engagement and reduce feelings of isolation or loneliness, bas...
- Dennis Thompson HealthDay Reporter
- |
- July 3, 2025
- |
- Full Page
Prevent 4th of July Firework Injuries by Taking These Simple Steps
Fireworks are the pinnacle of many Fourth of July celebrations, and while they can be festive and fun, they can also land you in the emergency room if you don’t take proper precautions, experts warn.
About 250 people a day wind up in the ER with fireworks-related injuries in the month before and after Independence Day. More than 75% ...
- Denise Mann HealthDay Reporter
- |
- July 3, 2025
- |
- Full Page
Want More Exercise? Go To Bed Earlier, Study Suggests
The age-old “early to bed, early to rise” proverb applies to your daily exercise regimen as well as your health, wealth and wisdom, a new study says.
Folks who get to bed earlier tend to be more physically active every day, researchers reported June 30 in the Proceedings of the National Academy of Sciences.
On av...
- Dennis Thompson HealthDay Reporter
- |
- July 3, 2025
- |
- Full Page
Smartphone-Controlled Nerve Stimulator Returns Golfer To The Links
Avid golfer Robert Knorr found he was no longer able to hit the links last year, due to neuropathy in his legs and feet.
The nerve pain got so bad that Knorr needed a cane — and sometimes a wheelchair — to get around.
But the 69-year-old retired oil company executive has traded that cane for his golf clubs, thanks to a sp...
- Dennis Thompson HealthDay Reporter
- |
- July 3, 2025
- |
- Full Page
Teen Drivers Spend A Fifth Of The Time Looking At Their Smartphone, Study Says
About a fifth of the time, a teenage driver is looking at their smartphone rather than the road or their rearview, a new study says.
Teen drivers spend an average 21% of each trip looking at their phone, according to results published today in the journal Traffic Injury Prevention.
Worse, these weren’t quick glances &m...
- Dennis Thompson HealthDay Reporter
- |
- July 3, 2025
- |
- Full Page
Schools Should Be Prepared For These Three Medical Emergencies
There are three common health emergencies for which all U.S. schools should be prepared, a new study says.
Brain-related crises like seizures, psychiatric conditions or substance abuse, and trauma-related injuries are the three main reasons paramedics respond to schools, according to a new report in the journal Pediatrics.
A...
- Dennis Thompson HealthDay Reporter
- |
- July 3, 2025
- |
- Full Page
Wildfire Smoke Linked To Heart Failure Risk
Wildfire smoke might increase a person’s risk of developing heart failure, a new study suggests.
People had a 1.4% higher risk of heart failure for every 1 microgram per cubic meter increase in their exposure to particle pollution from wildfires, researchers report in the Journal of the American College of Cardiology.
...
- Dennis Thompson HealthDay Reporter
- |
- July 3, 2025
- |
- Full Page
Could Dairy Be Causing Your Bad Dreams?
Having bad dreams after eating ice cream or cheese? Your stomach may be trying to tell you something.
New research shows that people with worse symptoms of lactose intolerance tended to report more frequent nightmares, NBC News reported.
The research, published July 1 in Frontiers in Psychology, looked at the eating...
- I. Edwards HealthDay Reporter
- |
- July 2, 2025
- |
- Full Page